Improve the symptoms of urinary incontinence with leva®
leva® is a new clinically proven, prescription-only treatment option for urinary incontinence, a serious medical condition that affects over 60% of your female members.1
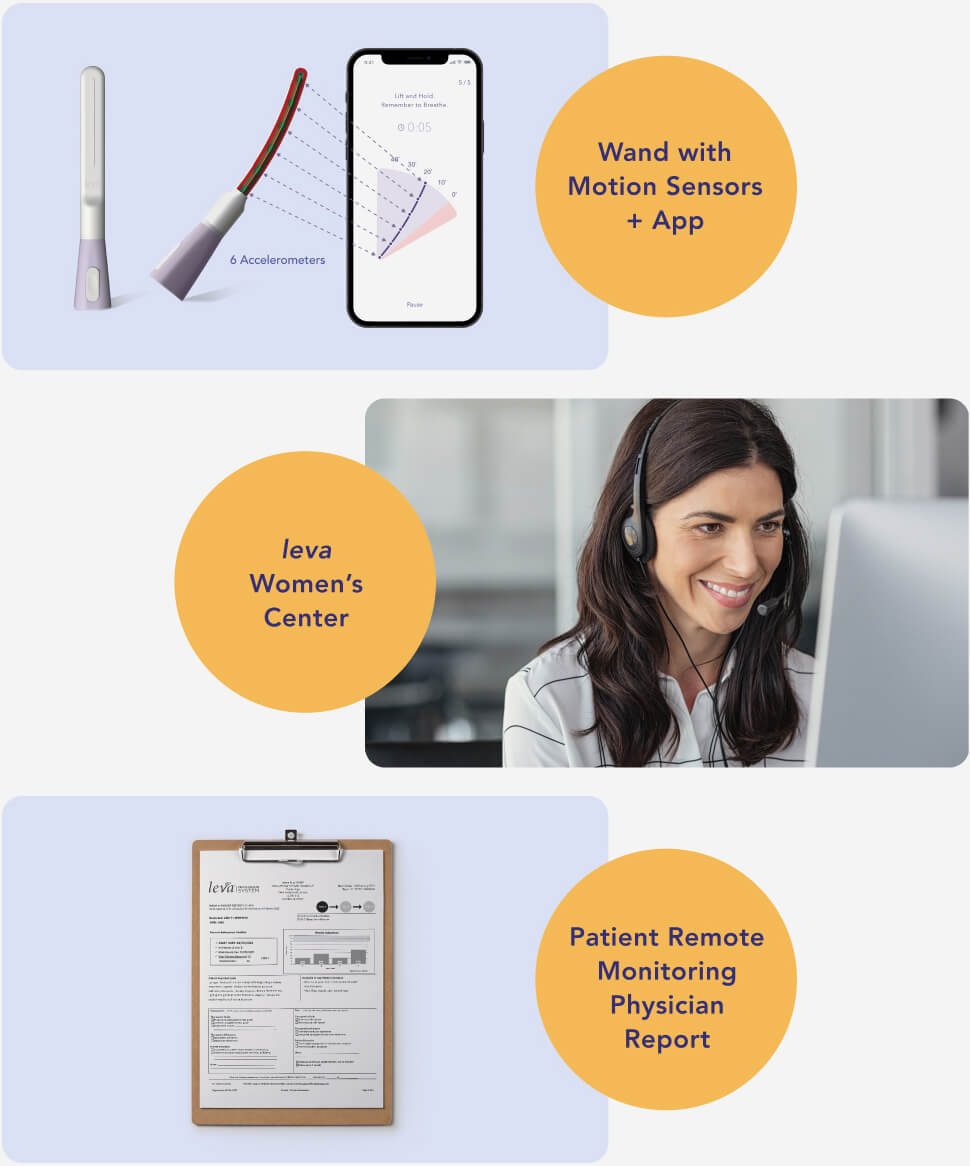
How leva® works
leva® is an at-home, easy-to-use software-driven pelvic health program that can help women strengthen their pelvic floor muscles and decrease their symptoms of urinary incontinence.
Wand with Motion Sensors + App
Inserted vaginally, the leva wand uses motion sensors to detect movement during pelvic floor muscle training, and communicates with the app to direct treatment and provide real-time feedback.
leva Women's Center
The leva system is supported by the leva Women's Center, a team of coaches who help patients with training, adherence and personal symptom goals.
Patient Remote Monitoring Physician Report
The report captures the adherence and symptom data that providers need to monitor patient progress over time.
Indication
The leva® Pelvic Health System is prescription-only and FDA-cleared for the treatment of stress, mixed and mild to moderate urgency urinary incontinence, including overactive bladder.
Prescription Reimbursement Identifier
60002099440

Clinical outcomes
Published in Obstetrics & Gynecology (“The Green Journal”), our pivotal 8-week randomized controlled trial (RCT) with 299 women demonstrates that pelvic floor muscle training guided by leva delivers better UI symptom improvement than Kegels alone.2 Primary outcomes were change in stress urinary incontinence (SUI) episodes on a 3-day bladder diary and change in Urogenital Distress Inventory (UDI-6) score.
- The 3-day bladder diary results showed that the median number of SUI episodes at 8 weeks was significantly fewer in the leva arm than in the Kegel arm. Quantitatively, leva users saw their leaking episodes decrease from nearly 2 leaks every day to 1 leak every 3 days.2
- For the (UDI-6) scores on symptom relief, the leva users reached a significantly greater score improvement compared to the Kegel arm. In fact, leva users saw their scores drop from 52.9 to 36.3, a level which is likely no longer bothersome to most patients.2,3
- The leva users saw statistically significant symptom improvement as early as 4 weeks into treatment.2
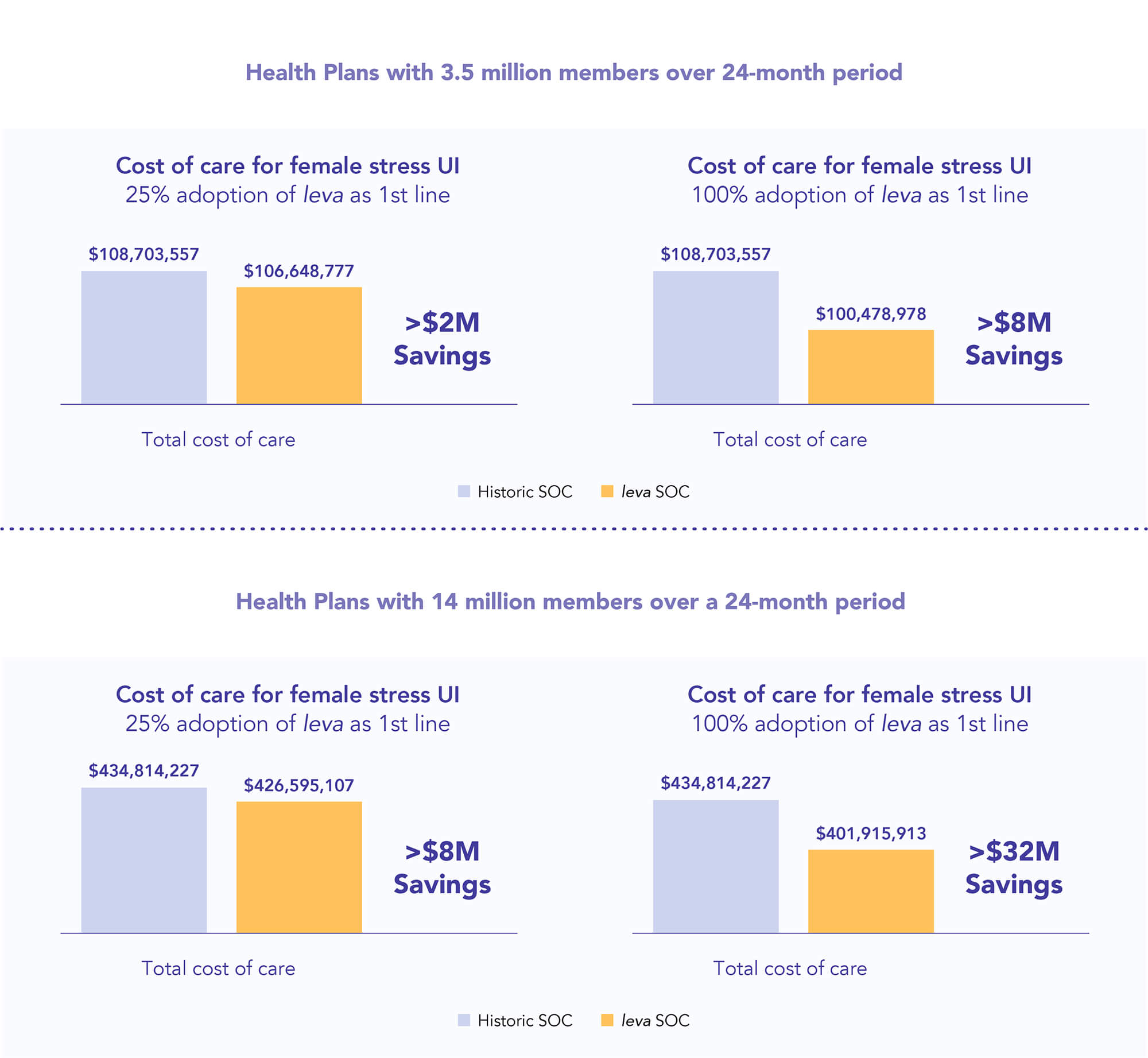
Plan savings with leva
Publications
Several publications support the finding that UI is costly and undertreated, and there is a real lack of documented first line care.
Resource Utilization/Cost to Payers
Conclusion: Women with SUI/MUI consume significantly higher medical resources and incur higher costs to health plans, compared to women without SUI/MUI. While reasons for this are not fully understood, improved and standardized treatment for women with SUI/MUI may positively affect cost and outcomes.
Status: Published; Neurourol Urodyn. 2022; 1-10. doi:10.1002/nau.24989
Care Pathway Adherence
Conclusion: Majority of SUI or MUI patients do not receive a behavioral intervention as their first-line treatment, which is a recommendation by professional society guidelines. This was found to affect the cost burden for health plans; those that were non-adherent had significantly higher costs 2-years post-index.
Status: Accepted; Neurourol Urodyn – publication date TBD
Utilization of Pelvic Floor PT
Conclusion: Percentage of women with a PT visit associated with an incident SUI or MUI diagnosis was low (2.6%), and 30% of this group completed three or more PT visits. This suggests poor adherence to clinical guidelines regarding supervised treatment of UI in women.4
Status: Published; Neurourol Urodyn. 2022; 1-8. doi:10.1002/nau.24913
Economic impact to the plan - Savings opportunity through offering improved first-line care for members with leva vs. historic standard of care (SOC)*
*PFMT supervised by a PT or unsupervised Kegels
Examples
Key Takeaways
Cost savings with leva within UI standard of care
- Majority of savings from decreased utilization of advanced treatments
- Improved care and systematic cost savings
Increased utilization of leva should lead to lower costs for health plans
Interested in learning more about urinary incontinence?
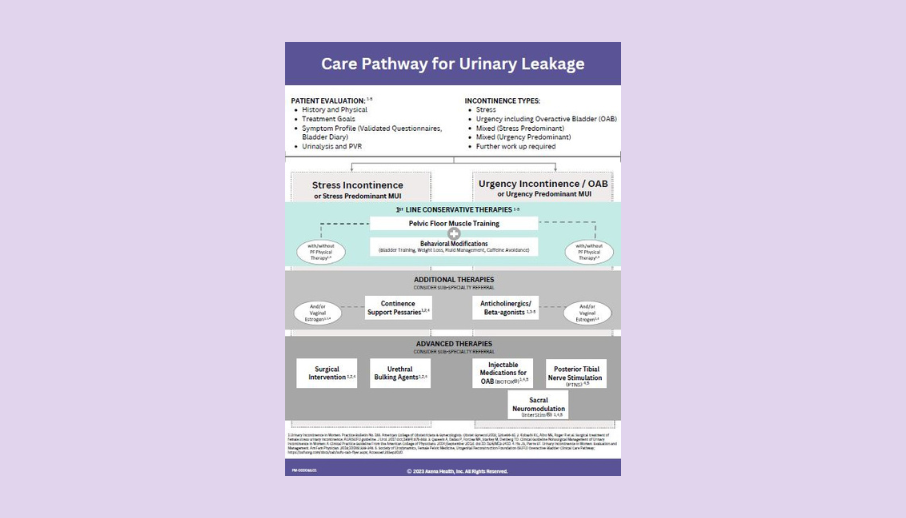
Care Pathway
Use this tool to help you discuss UI treatment options and the role that pelvic floor muscle training plays in treatment/pelvic health.
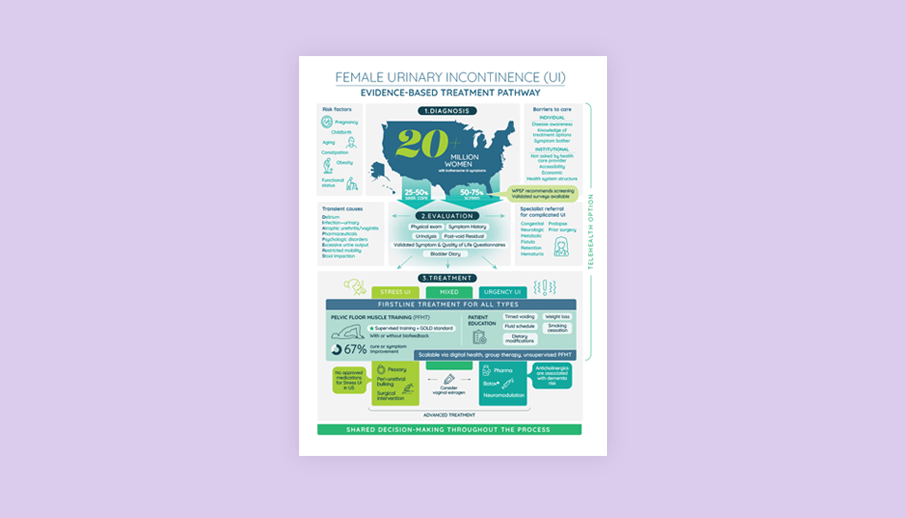
Evidence-Based Treatment Pathway Infographic
Download this infographic to support education and shared decision-making in screening, evaluation, and treatment for female urinary incontinence.

Evidence-Based Treatment Pathway Publication
Read the full study on the prevalence of female urinary incontinence and how it can be treated.
Contact us today to learn about the benefits of leva
References
- Patel UJ, Godecker AL, Giles DL, Brown HW. Updated Prevalence of Urinary Incontinence in Women: 2015-2018 National Population-Based Survey Data [published online ahead of print, 2022 Jan 12]. Female Pelvic Med Reconstr Surg. 2022;10.1097/SPV.0000000000001127. doi:10.1097/SPV.0000000000001127
- Weinstein, MM, Dunivan, G, Guaderrama, NM, Richter, HE. Digital Therapeutic Device for Urinary Incontinence: A Randomized Controlled Trial [published online ahead of print, 2022 March 10]. Obstet Gynecol. doi: 10.1097/AOG.0000000000004725.
- Sanderson DJ, Zavez A, Meekins AR, Eddib A, Lee TG, Barber MD, Duecy E. The Patient Acceptable Symptom State in Female Urinary Incontinence. Female Pelvic Med Reconstr Surg. 2022 Jan 1;28(1):33-39. doi: 10.1097/SPV.0000000000001055. PMID: 34009829.
- McKinney, JL, Datar, M, Pan, L-C, Goss, T, Keyser, LE, Pulliam, SJ. Retrospective claims analysis of physical therapy utilization among women with stress or mixed urinary incontinence. Neurourol Urodyn. 2022; 1- 8. doi:10.1002/nau.24913
Important Safety Information
The Leva® Pelvic Health System is intended for: (i) strengthening of the pelvic floor muscles; (ii) rehabilitation and training of weak pelvic floor muscles for the treatment of stress, mixed and mild to moderate urgency urinary incontinence (including overactive bladder) in women; and (iii) rehabilitation and training of weak pelvic floor muscles for the first-line treatment of chronic fecal incontinence (>3-month uncontrolled passage of feces) in women. This product interacts with the user via smartphone technology.
Important Safety Information for the Leva Pelvic Health System: Do not share the Leva system. Leva is a single-user medical device. Do not use the Leva Pelvic Health System while pregnant, or if you think you may be pregnant, unless authorized by your healthcare provider. Do not leave the Leva vaginal wand in your body for longer than necessary to complete the training session. Remove the vaginal wand after each training session. Do not use the Leva Pelvic Health System in any other place in your body. Do not have sexual intercourse while the vaginal wand is inserted. If you experience odor, fever, vomiting, diarrhea, any signs of infections or any flu-like symptoms, contact your doctor immediately. If you experience redness or swelling near the insertion area, or signs of an allergic reaction, contact your healthcare provider immediately For a complete summary of the risks and instructions for the Leva System, see its Instructions for Use available at www.levatherapy.com. Treatment with the Leva Pelvic Health System is prescribed by your healthcare provider. This treatment is not for everyone. Please talk to your healthcare provider to see if it is right for you. Your healthcare provider should discuss all potential benefits and risks with you.
Limited Warranty
Axena Health warrants the leva® Pelvic Health System (“leva” or the “device”) against defects in materials and workmanship for six (6) months from the date of original purchase. Alleged defects must be reported to Axena Health by the original purchaser prior to the end of the warranty. Warranty claims must be supported with reasonable evidence of the claimed defect. Axena Health may require the device be returned and if required, will provide customer with a Return Material Authorization number, which must be displayed on the outside packaging sent to Axena Health.
Axena Healthreserves the right to determine whether to repair or replace a device determined to be defective. In the event of replacement, the warranty period of the replacement device will be run from the date of the original purchase. Repair or replacement of your device will not extend the time period of this limited warranty.
This limited warranty does not cover cosmetic or battery deterioration or damage caused by excessive wear and tear, physical or natural destruction, accident, misuse, neglect or other external causes. Any attempt to disassemble the device will invalidate this limited warranty and any use of the device that is contrary to its Instructions for Use (available on Axena Health ’s website) may invalidate this limited warranty.
REPAIR OR REPLACEMENT OF THE DEVICE OR ITS AFFECTED COMPONENT ARE THE EXCLUSIVE REMEDIES PROVIDED IN CONJUNCTION WITH THE PURCHASE AND USE OF THE DEVICE. TO THE EXTENT PERMITTED BY LAW, THIS WARRANTY AND THE REMEDIES SET FORTH ARE EXCLUSIVE AND IN LIEU OF ALL OTHER WARRANTIES, REMEDIES AND CONDITIONS, WHETHER ORAL, WRITTEN, STATUTORY, EXPRESS OR IMPLIED. AXENA HEALTH DISCLAIMS ALL STATUTORY AND IMPLIED WARRANTIES, INCLUDING WITHOUT LIMITATION, WARRANTIES OF MERCHANTABILITY AND FITNESS FOR A PARTICULAR PURPOSE AND WARRANTIES AGAINST HIDDEN OR LATENT DEFECTS. IN SO FAR AS SUCH WARRANTIES CANNOT BE DISCLAIMED, AXENA HEALTH LIMITS THE DURATION AND REMEDIES OF SUCH WARRANTIES TO THE DURATION OF THIS EXPRESS WARRANTY AND, AT AXENA HEALTH ’S OPTION, REPAIR OR REPLACEMENT AS DESCRIBED ABOVE. SOME STATES DO NOT ALLOW CERTAIN WARRANTY LIMITATIONS, SO THE LIMITATION DESCRIBED ABOVE MAY NOT APPLY TO YOU.
Please contact Axena Healthto confirm the terms of this Limited Warranty prior to purchase. Axena Healthreserves the right, at its discretion, to modify its terms at any time prior to your purchase.