leva® is shown to be statistically superior to Kegels alone
At 8 weeks, leva users experienced better symptom improvement for stress and stress-dominant urinary incontinence compared to Kegels alone.1
leva users saw a decrease in leaking episodes from
nearly 2 leaks per day
to about 2 leaks per week
Symptom Improvement — Urinary Distress Inventory-6 (UDI-6) Score Change
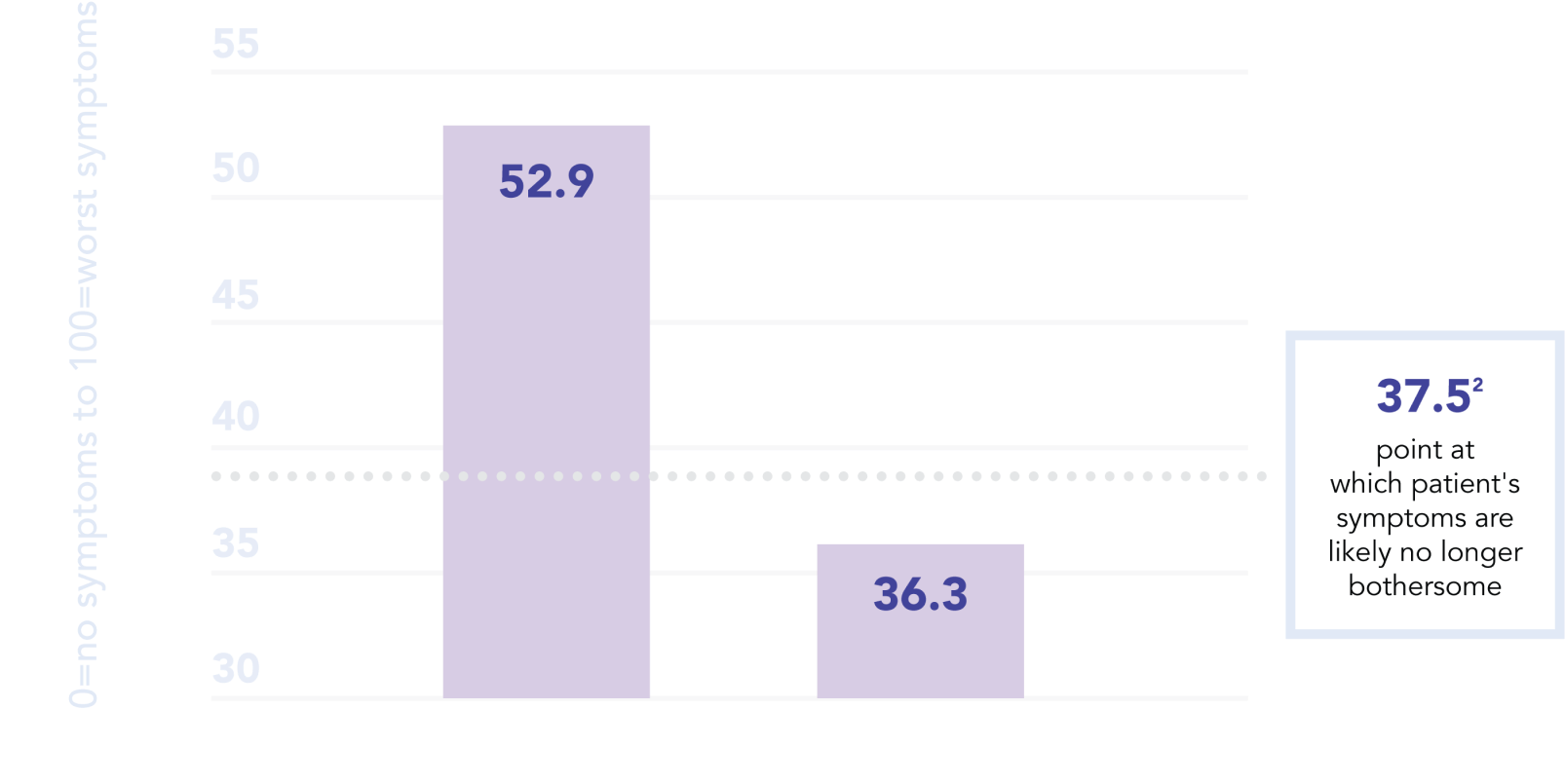
leva users saw
significant symptom improvement
as early as 4 weeks into treatment, according to UDI-6 symptom scores1
Help your patients take control of their pelvic health.
Contact our Provider Support Helpline to learn more.
Contact UsReferences
- Weinstein, MM, Dunivan, G, Guaderrama, NM, Richter, HE. Digital Therapeutic Device for Urinary Incontinence: A Randomized Controlled Trial [published online ahead of print, 2022 March 10]. Obstet Gynecol. doi: 10.1097/AOG.0000000000004725.
- Sanderson DJ, Zavez A, Meekins AR, Eddib A, Lee TG, Barber MD, Duecy E. The Patient Acceptable Symptom State in Female Urinary Incontinence. Female Pelvic Med Reconstr Surg. 2022 Jan 1;28(1):33-39. doi: 10.1097/SPV.0000000000001055. PMID: 34009829.
Important Safety Information
The Leva® Pelvic Health System is intended for: (i) strengthening of the pelvic floor muscles; (ii) rehabilitation and training of weak pelvic floor muscles for the treatment of stress, mixed and mild to moderate urgency urinary incontinence (including overactive bladder) in women; and (iii) rehabilitation and training of weak pelvic floor muscles for the first-line treatment of chronic fecal incontinence (>3-month uncontrolled passage of feces) in women. This product interacts with the user via smartphone technology.
Important Safety Information for the Leva Pelvic Health System: Do not share the Leva system. Leva is a single-user medical device. Do not use the Leva Pelvic Health System while pregnant, or if you think you may be pregnant, unless authorized by your healthcare provider. Do not leave the Leva vaginal wand in your body for longer than necessary to complete the training session. Remove the vaginal wand after each training session. Do not use the Leva Pelvic Health System in any other place in your body. Do not have sexual intercourse while the vaginal wand is inserted. If you experience odor, fever, vomiting, diarrhea, any signs of infections or any flu-like symptoms, contact your doctor immediately. If you experience redness or swelling near the insertion area, or signs of an allergic reaction, contact your healthcare provider immediately For a complete summary of the risks and instructions for the Leva System, see its Instructions for Use available at www.levatherapy.com. Treatment with the Leva Pelvic Health System is prescribed by your healthcare provider. This treatment is not for everyone. Please talk to your healthcare provider to see if it is right for you. Your healthcare provider should discuss all potential benefits and risks with you.
Limited Warranty
Axena Health warrants the leva® Pelvic Health System (“leva” or the “device”) against defects in materials and workmanship for six (6) months from the date of original purchase. Alleged defects must be reported to Axena Health by the original purchaser prior to the end of the warranty. Warranty claims must be supported with reasonable evidence of the claimed defect. Axena Health may require the device be returned and if required, will provide customer with a Return Material Authorization number, which must be displayed on the outside packaging sent to Axena Health.
Axena Healthreserves the right to determine whether to repair or replace a device determined to be defective. In the event of replacement, the warranty period of the replacement device will be run from the date of the original purchase. Repair or replacement of your device will not extend the time period of this limited warranty.
This limited warranty does not cover cosmetic or battery deterioration or damage caused by excessive wear and tear, physical or natural destruction, accident, misuse, neglect or other external causes. Any attempt to disassemble the device will invalidate this limited warranty and any use of the device that is contrary to its Instructions for Use (available on Axena Health ’s website) may invalidate this limited warranty.
REPAIR OR REPLACEMENT OF THE DEVICE OR ITS AFFECTED COMPONENT ARE THE EXCLUSIVE REMEDIES PROVIDED IN CONJUNCTION WITH THE PURCHASE AND USE OF THE DEVICE. TO THE EXTENT PERMITTED BY LAW, THIS WARRANTY AND THE REMEDIES SET FORTH ARE EXCLUSIVE AND IN LIEU OF ALL OTHER WARRANTIES, REMEDIES AND CONDITIONS, WHETHER ORAL, WRITTEN, STATUTORY, EXPRESS OR IMPLIED. AXENA HEALTH DISCLAIMS ALL STATUTORY AND IMPLIED WARRANTIES, INCLUDING WITHOUT LIMITATION, WARRANTIES OF MERCHANTABILITY AND FITNESS FOR A PARTICULAR PURPOSE AND WARRANTIES AGAINST HIDDEN OR LATENT DEFECTS. IN SO FAR AS SUCH WARRANTIES CANNOT BE DISCLAIMED, AXENA HEALTH LIMITS THE DURATION AND REMEDIES OF SUCH WARRANTIES TO THE DURATION OF THIS EXPRESS WARRANTY AND, AT AXENA HEALTH ’S OPTION, REPAIR OR REPLACEMENT AS DESCRIBED ABOVE. SOME STATES DO NOT ALLOW CERTAIN WARRANTY LIMITATIONS, SO THE LIMITATION DESCRIBED ABOVE MAY NOT APPLY TO YOU.
Please contact Axena Healthto confirm the terms of this Limited Warranty prior to purchase. Axena Healthreserves the right, at its discretion, to modify its terms at any time prior to your purchase.